Pharm Exec’s latest report covers the significant transformations taking place in the global biopharma industry, with the trajectory of commercial output and product direction taking centre stage. In this blog, we will explore the key trends and factors shaping the industry's future, including the impact of non-pandemic treatment trends, market drivers, and other macroeconomic factors. We will also delve into the implications of mergers and acquisitions (M&A) and the ongoing challenges posed by the COVID-19 pandemic.
A shifting scene
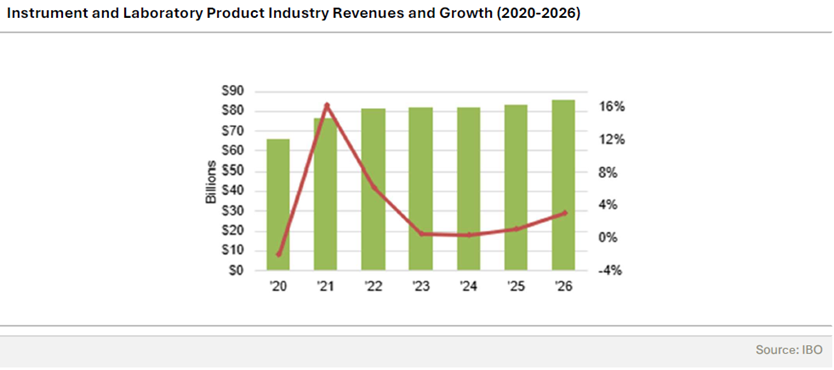
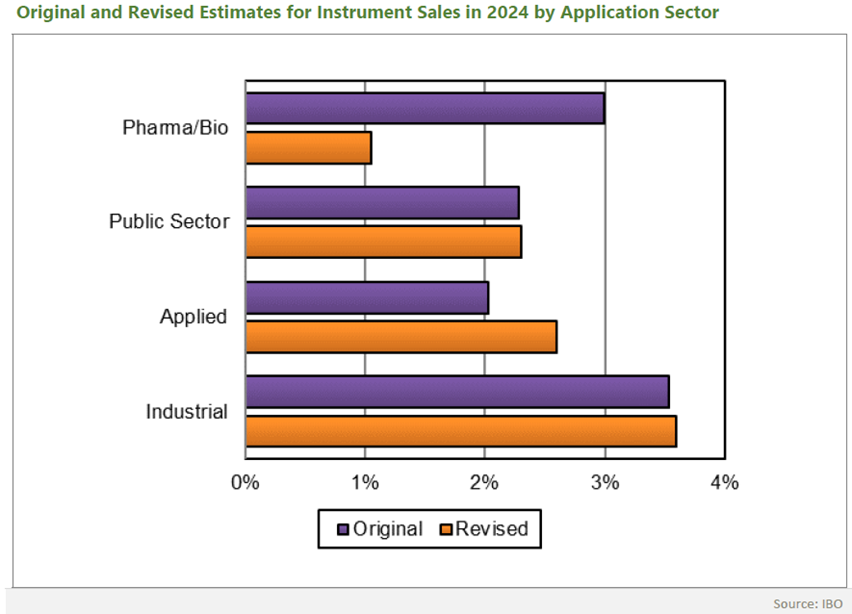
The biopharma industry is on the cusp of a significant shift, and whilst the numbers reflect the most recent full-year drug revenue performance in 2022, they also hint at a changing landscape driven by emerging treatment trends and market dynamics. According to the report, in the coming years, we can expect a reshuffle, with new players entering the scene and established companies facing potential challenges.
Emerging treatments
With the global obesity market projected to reach close to $50 billion by 2028,[i] there is a tremendous growth opportunity in weight loss treatments. This growth and increased opportunity will drive improvements to biopharmaceutical treatments. Other treatments include respiratory syncytial virus (RSV) vaccines, therapies for Alzheimer's disease, new antibody-drug conjugates (ADCs) for cancer, and more effective options in chronic obstructive pulmonary disease (COPD), geographic atrophy (GA), and chronic inflammatory demyelinating polyneuropathy (CIDP).
Navigating business pressures
Biopharma companies are not immune to current business pressures, and like many others, the industry is grappling with wider inflation and supply chain challenges that impact operations and profitability. Mid-range companies are seeking opportunities to ascend and join their larger peers, while smaller players are aiming to make their mark. However, these aspirations must be balanced with the tough operational choices that many companies, both large and small, have been forced to make, ranging from layoffs to pipeline and business unit cuts.
The impact of M&A
M&A has long been a driving force in the biopharma industry, however, recent developments, such as the US Federal Trade Commission's (FTC) lawsuit blocking the anticipated acquisition of Horizon Therapeutics by Amgen, have created a sense of caution among industry players. The FTC's move highlights growing scrutiny of potential ‘mega mergers’, as regulators aim to assess the impact on competition. As a result, we may see more deals between mid-range companies, creating increased competition for the top-tier players. However, despite potential uncertainties surrounding M&A, the biopharma industry remains optimistic about the potential for deals in the coming years. Investment banking company, Stifel, predicts that biopharma M&A dollar volume could reach $208 billion by the end of the year.[ii] Ultimately, companies considering M&A must navigate antitrust concerns and ensure that any proposed deals align with their long-term strategic goals.
Pricing pressures and patent expirations
The Inflation Reduction Act in the US, passed in August 2022, has paved the way for Medicare price negotiations, putting pressure on companies to navigate these waters effectively. Pfizer, Roche, GSK, Merck and Co., and Bristol Myers Squibb (BMS) are among the companies under particular scrutiny as they seek to sustain their pipelines and mitigate the impact of near and long-term patent expirations. These challenges have prompted a renewed focus on strategic partnerships, acquisitions, and pipeline diversification.
COVID-19's impact and the road ahead
The COVID-19 pandemic has had a profound impact on the biopharma industry, both positive and negative, according to the report. Pfizer experienced significant growth due to the success of its COVID messenger RNA (mRNA) vaccine, Comirnaty, and its antiviral therapy, Paxlovid. However, the drop in demand for coronavirus products and testing has already affected players across the board. Companies are now shifting their focus from selling vaccines and drugs directly to governments to targeting healthcare providers. This shift, coupled with the anticipated decline in COVID-related revenues, necessitates a proactive approach to replenish losses.
Conclusion
The global biopharma industry is undergoing a transformative phase, driven by changing treatment trends, market dynamics, and regulatory pressures. As the industry navigates the evolving market forces, it is imperative for companies to adapt and embrace new opportunities. Strategic partnerships, innovative research and development, and a focus on patient-centric solutions will be key to thriving in the changing landscape. The future of the biopharma industry lies in the hands of those who can effectively balance business pressures, regulatory compliance, and the pursuit of scientific innovation.
How can we help you navigate the change?
Our close collaboration with clients spans various life science, technology, and industrial sectors. We specialise in supporting clients to communicate effectively with complex audiences across China, North America, and Europe.
If you’d like to explore how we can assist your life science company in navigating the evolving life science landscapes, why not contact us and find out how we can support.
References
[i] Morgan Stanley Research, August 2022. Available at: https://www.morganstanley.com/ideas/obesity-drugs-investment-opportunity
[ii] Christel, Michael. Pharmaceutical Executive: Volume 43, Issue 6. June 2023